It May Take Time For Biologic Drugs To Take Effect
When you start taking an anti-TNF drug, it may take up to 8 weeks to notice any improvement in your UC symptoms, according to the Crohns and Colitis Foundation. While some people notice an immediate improvement, the possibility of a delayed effect means youll need to continue your treatment for several weeks before concluding that it isnt effective, if that turns out to be the case.
Side Effects And Interactions
The more common side effects of biologics can include:
Biologic drugs may interact with other drugs and biologic agents, including:
- natalizumab , which can be used to treat Crohns disease or multiple sclerosis
- tocilizumab , anakinra , abatacept , which are primarily used to treat arthritis
- theophylline , an asthma medication
- live vaccines such as the varicella zoster vaccine
Integrin Receptor Antagonists For Ulcerative Colitis:
These are the biologics that have the same role of reducing inflammation just like anti-TNF but they work differently.
These are also antibodies just like the anti-TNF but they are against the integrin molecules. They bind to the alpha 4 subunit of the integrin molecule resulting in halting of the movement of lymphocytes or cytokines.
The two adhesion molecules that are overexpressed in patients with inflammatory bowel diseases including ulcerative colitis are ICAM-1 and MAdCAM-1.
These two molecules are targets for the commonly used drug, Vedolizumab , and the new drug, Ertolizumab.
The effect is reduced inflammation due to a lack of movement of cells responsible for inflammation and thus the symptoms of UC are relieved.
Read Also: How Do You Treat A Duodenal Ulcer
Things To Know About Biologics For Ulcerative Colitis
Biologics are one option in the treatment of ulcerative colitis. Heres what you should know before starting biologic therapy.
If you have moderate to severe ulcerative colitis and aren’t responding to or can’t tolerate traditional medications, such as aminosalicylates or immunomodulators, your doctor may ask you to start taking a biologic.
Biologic drugs, such as adalimumab and infliximab, can help stop inflammation and keep your disease in remission if other UC medications arent effective. But these drugs are also complex and may not work for everyone, so its important to learn more about them if youre new to biologic therapy.
Here are 10 key facts about biologic drugs for UC.
Prognostic Testing: The Start Of Precision Medicine In Ibd

Being able to effectively predict the course of IBD at the time of diagnosis to identify an individual patients risk of experiencing frequently relapsing disease would allow those patients at high-risk to be prescribed biologics earlier, which could improve their chance of going into remission and reduce the probability of disease-related complications.
Although clinical criteria have been reported to associate with outcome, these perform poorly when tested, making it difficult to identify high-risk patients. Recent years have brought important insights into the biology underlying different disease course in IBD patients, in particular the understanding of the impact of the immunological state known as T-cell exhaustion12,13. This has allowed the development of PredictSURE IBDTM, a fully validated, CE marked whole blood prognostic test for Crohns disease and ulcerative colitis14. Using qPCR, PredictSURE IBD measures the expression of 17 genes and a proprietary algorithm to sort patients into high- and low-risk subgroups.
Read Also: How To Ease Stomach Ulcer Pain
You May Like: Stelara Ulcerative Colitis Side Effects
The Importance Of Balancing Other Risks
While medication risk is an important consideration that is well-recognized, it is important to recognize the higher risks of complications from poorlycontrolled disease activity, including fistula, stricture, and surgery. Notably, Osterman et al. showed that higher disease activity and corticosteroid use were associated with an increased risk of infection.95 Furthermore, the increased mortality risk associated with corticosteroids and narcotics has been well demonstrated.96-98 Lastly, recent studies have shown the 10-year risk of surgery is around 40% for CD99 and around 15% for UC.100 In patients with CD, the risk of developing an intestinal complication, such as fistula or stricture, is 50% within 20 years after diagnosis.101 Thus it is important to put the risks of medications into perspective with the high risks of poorly-controlled IBD.
Entyvio Should Be First Choice For Patients With Uc Who Previously Failed Remicade
We were unable to process your request. Please try again later. If you continue to have this issue please contact .
In a direct comparison with Humira, Entyvio won out as the top choice for second line therapy for patients with ulcerative colitis who failed therapy with Remicade, according to research published in Inflammatory Bowel Diseases.
Massimo Claudio Fantini, MD, PhD, from the University of Rome, and colleagues wrote that there are no solid data to support which drug should be used as a follow-up in patients who do not respond or lose response to Remicade .
At the moment, there is no clear indication on the most appropriate second-line biological therapy in case of failure to , and in the absence of head-to-head comparative efficacy data, the choice between a second anti-TNF-alpha or the anti-integrin is often based on clinicians personal experience, drug availability, and economic issues, they wrote.
Researchers analyzed clinical records of 161 patients with UC who failed infliximab therapy and were candidates to receive either Humira or vedolizumab. The primary endpoint was therapeutic failure at week 52, and secondary endpoints included therapy discontinuation at weeks 8, 24 and 52, discontinuation-free survival and safety.
Patients in the study were either primary or secondary infliximab failures or infliximab intolerants . Researchers determined that 64 patients received adalimumab and 97 patients received vedolizumab .
Also Check: What Are The Signs And Symptoms Of Ulcer
The Advantages Of Biologics For Ulcerative Colitis
Biologics can improve gut symptoms, bringing about and maintaining remission in people with moderate to severe ulcerative colitis. They can also reduce the need for hospitalization and surgery, says the British organization Crohn’s & Colitis UK.
One key advantage of biologic therapies over other types of treatment for ulcerative colitis is that their mechanisms of action are more precisely targeted to the factors responsible for the condition, notes the Crohns & Colitis Foundation.
Unlike corticosteroids, for example, which affect the whole body and may produce major side effects, biologic agents act more selectively. These therapies are targeted to particular proteins that have already been proven to be involved in ulcerative colitis.
Patients are often nervous about biologics, says Laura Raffals, MD, a gastroenterologist at the Mayo Clinic in Rochester, Minnesota. But theyre not as scared about taking a corticosteroid, and thats an important conversation to have because the data shows that patients on biologics are much safer and stay in remission longer. The serious side effects we see are from steroids or narcotics, not biologics.
What Differences Might There Be
Biosimilars are thoroughly tested. They meet strict standards to show they are as safe, effective and have no clinically meaningful differences from the originator. Where NICE has recommended the use of a biological medicine, they state that the same guidance applies to the biosimilar.However, there are likely to be some small differences including:
Some people may be sensitive to latex, which is used as a needle cover in some of some types of biosimilars, or citrate which is sometimes included as one of the ingredients.Some types of adalimumab are available without citrate or latex, and you can ask to try one of these.
Dont Miss: Is Milk Good For Ulcers
Read Also: How To Check For Ulcerative Colitis
Who Should Try Biologics
Biologics are recommended to UC patients with moderate to severe disease, often after other treatments have failed, says Neilanjan Nandi, M.D., an associate professor of gastroenterology and hepatology at Drexel University in Philadelphia. Oral steroids are typically tried first, according to the American Academy of Family Physicians , but if they dont have the desired response, biologics are often the next step. People with milder UC are typically first treated with drugs called 5-aminosalicylic acids, including , which also help reduce inflammation.
“However, UC patients that have more than four to six daily bowel movements may have too much inflammation for mesalamines to have enough healing effect, explains Dr. Nandi. The chronic blood loss can result in iron deficiency or , and the patient’s quality of life may likely suffer from chronic fatigue and distress.
“Quality of life” can mean a lot of things, but in this case, Dr. Nandi says its assessed based on clinical symptoms and complications, the impact they’re having on the persons , and how dependent they are on steroids. (Long-term steroid use can lead to impaired healing, increased blood pressure, and osteoporosis, according to the CCF. Risk of infections is one of the most important complications which is often worse than biologic medications.
If there’s a significant impact, then a doctors will usually recommend trying a biologic.
Prednisone Prednisolone And Methylprednisolone
Prednisone is taken by mouth and is available as:
- an immediate-release tablet
- a delayed-release tablet
- a liquid solution
Its available as a generic drug and as the brand-name drugs Prednisone Intensol and Rayos .
The forms of prednisolone that are FDA approved for UC are:
- immediate-release tablet
- liquid solution
You can take any of these forms by mouth. Prednisolone is available as a generic drug and as the brand-name drugs Millipred and Prelone .
Methylprednisolone comes in two forms:
- injectable medication
Its available as a generic drug and as the brand-name drugs Medrol and Depo-Medrol .
Side effects, complications, and interactions
When given in high doses, the side effects of these drugs are similar. The more common side effects can include:
- increased blood sugar levels
Immunomodulators are drugs that decrease the bodys response to its own immune system. The result is lowered inflammation throughout your body.
Immunomodulators may reduce the number of UC flare-ups you have and help you stay symptom-free longer.
Theyre generally prescribed to people whose symptoms havent been managed with 5-ASA drugs and corticosteroids. However, these drugs may take several months to start working.
The FDA has not approved immunomodulators for the treatment of UC.
However, theyre well supported in medical literature as useful options, and your doctor may still prescribe them. This is known as off-label drug use.
Recommended Reading: Types Of Steroids For Ulcerative Colitis
Interleukin Inhibitors For Ulcerative Colitis:
Interleukin inhibitors are the biologics used to suppress the immune system. Interleukins of different types are responsible for the proper functioning of the immune system as well as the movement of immune cells. In the case of UC, they target the proteins that direct inflammation.
It is considered one of the most effective treatments with the infusion of a single dose which is followed by another dose after 8 weeks.
A New Indication Of Success: Fda Approves Ozanimod For Ulcerative Colitis
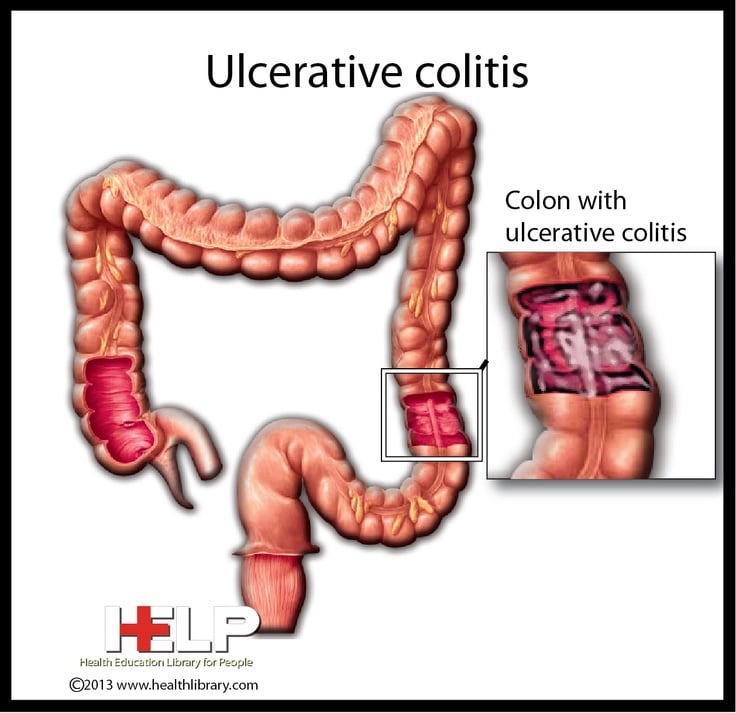
The novel drug created at Scripps Research has achieved a second FDA approval, this time for ulcerative colitis, as clinical trials continue for Crohns disease.
May 27, 2021
LA JOLLA, CAOzanimod, the drug invented at Scripps Research that won FDA approval last year for relapsing forms of multiple sclerosis, has been approved in the United States for a second high-need medical condition, ulcerative colitis.
The once-daily oral drug, sold by Bristol Myers Squibb under the name Zeposia, can now be prescribed to treat adults with moderate to severe forms of the inflammatory bowel disease. Notably, its the first drug in a novel class of immune-modulating compounds to be approved for ulcerative colitis, which affects about 1 million people in the United States.
For patients with ulcerative colitis, this oral drug offers a better and more convenient option to control disease progression and improve quality of life, says Hugh Rosen, MD, PhD, who invented ozanimod along with fellow Scripps Research professor Edward Roberts, PhD, and their laboratory colleagues. The hope is that this will lead to fewer dangerous complications or serious infections than current treatment options, providing a steadier path for newly diagnosed patients as well as those failing other treatments.
Additional molecules developed by Rosen and Roberts at Scripps Research are currently in phase 2 clinical trials for major depressive disease and anxiety, and phase 1 studies for treatment of autism.
Read Also: What Happens When You Have A Bleeding Ulcer
Interactions With Other Medications
Interactions between biologics and other medications can be significant, and each type of biologic drug can present different risks.
A doctor should be aware of all the over-the-counter or prescription medications, and all the supplements, herbal medications, and vitamins that a person takes before they prescribe a biologic.
Anyone using a biologic for UC should speak with a doctor before receiving a vaccine. For people who use certain biologic medications, including golimumab, infliximab, and adalimumab, having a live vaccine can be dangerous, and doctors recommend avoiding it.
$10,00030,000 per year on average, and the more expensive types can cost more than $500,000 annually.
Although many medical insurance plans may cover biologics, depending on a persons deductible, the cost can still be high.
A doctor may instead recommend a type of drug they call a biosimilar. There is very little clinical difference between these drugs and biologics. Biosimilars are less expensive but just as safe and effective.
The FDA approved the use of biosimilars in an effort to reduce costs. However, the FDA does not regulate whether insurance companies cover the costs of these drugs.
The following table provides an overview of biologics available to treat UC. The abbreviation IV stands for intravenous.
| Drug |
A range of biologic drugs can reduce UC symptoms in adults and help achieve remission. For children with moderate to severe UC, a doctor may prescribe infliximab.
How Long Do Biologics Take To Work
Biologic therapy can take time to start working. Some people living with Crohn’s disease or ulcerative colitis experience relief soon after their first dose. Depending on the medication, it can take several weeks to notice an improvement in symptoms. It is important to stick to your treatment plan and give your biologic medication time to take effect.
Several MyCrohnsAndColitisTeam members have shared questions and experiences about how long it takes biologic drugs to work. One member asked, “Has anyone been on Humira? How long does it take to work?” In response, another member offered, “I’ve been on Humira for eight months. It worked for me right away.”
Read Also: Why Is Ulcerative Colitis Worse At Night
Humira Pen And Other Forms
Humira comes in three forms: a prefilled pen, a prefilled syringe, and a vial of liquid solution. The drug is given as a subcutaneous injection. A healthcare professional can give you Humira injections. But you may be able to give yourself injections at home with the pen or syringe if your doctor approves. Youd need to be trained first.
Both the pens and syringes are prefilled with a single dose of Humira and come with needles. There are also special Humira starter packs available that contain several prefilled pens or syringes.
Humira also comes in a single-dose vial. But this form can only be used by healthcare professionals. You wont be able to use vials yourself.
If you have questions about which form of Humira is right for you or how to give yourself injections, talk with your doctor.
You May Like: 8 Foods To Eat During An Ulcerative Colitis Flare
Treating Moderate To Severe Ulcerative Colitis With Biologics
Treating ulcerative colitis has two main goals: helping to subdue your flare-up and maintaining it so you have fewer flare-ups. Biologics are among the newest drugs available for treating ulcerative colitis. They can be especially helpful for those who havent found relief yet.
Also Check: Why Do You Lose Weight With Ulcerative Colitis
Will Biosimilars Give Greater Access To Biologics In Ibd
The introduction of biosimilarsâ medicines that are very similar to an original biologic therapy which has come off patent â has increased competition in the biologics market. An increase in competition has resulted in a decrease in the cost of this type of therapy, the most expensive in inflammatory bowel disease . So, does this mean more inflammatory bowel disease patients will be prescribed biologics at an earlier stage in their treatment? We take a look.
Donât Miss: How To Cure Gastritis And Ulcers Naturally
Who Needs This Medication
Some doctors recommend biologics before trying prednisone because of the risk of becoming “steroid dependent”unable to stop prednisone without a flare-up in symptoms. But other doctors point out that not enough is known about the long-term benefits and side effects of starting out with a biologic.
It is the case that most studies of biologics have involved people who didn’t find relief with other medication, so we don’t know very much about the pros and cons of starting on a biologic before other medications. Biologics and prednisone provide quick relief, but biologics can be used for longer periods of time, whereas prednisone should only be used for short periods.
One strategy that doctors sometimes use is to have a patient begin using a biologic or prednisone for immediate relief and start azathioprine or mercaptopurine at the same time to prevent recurrent symptoms. In studies, the combination of the biologic infliximab and azathioprine provided better clinical symptoms relief than either medication alone, although patients reported that their quality of life was similar with combination or single medication use.
Don’t Miss: Prednisone For Ulcerative Colitis Reviews