If You Use More Than The Recommended Amount Of Stelara
Call your doctor right away if you believe youve used too much Stelara. Another option is to call the American Association of Poison Control Centers at 800-222-1222 or use its online tool. If you have severe symptoms, immediately call 911 or your local emergency number, or go to the nearest emergency room.
Do Stelara And Humira Work The Same Way
Stelara and Humira both work by suppressing overactive parts of your immune system. This helps lower inflammation that causes tissue damage and the symptoms of your condition. But they dont work in exactly the same way.
Heres how these drugs compare:
- Stelara lowers inflammation by blocking interleukin-12 and interleukin-23. These are inflammatory substances that your immune system makes in response to antigens . If your immune system overproduces these substances, it causes too much inflammation that can damage healthy tissues. This damage brings about symptoms of your condition.
- Humira lowers inflammation by blocking tumor necrosis factor .TNF is another substance that your immune system makes that leads to inflammation. It signals other immune cells so they can help fight infections. If your immune system makes too much TNF, it can cause too much inflammation that damages healthy tissues. This damage causes symptoms of your condition.
What To Expect During Treatment
The first time you receive Stelara, it will be given through an IV infusion at a doctors office or clinic. The amount given in your first dose will be determined by your weight.
After your first IV dose, you take Stelara as an injection at home. You can self-inject or have a friend or loved one help you.
Stelara comes in a prefilled syringe with a plunger top. No matter what your initial infusion dose was, your ongoing doses will be 90 milligrams. No measuring is necessary.
The needle cover on the syringe contains latex, so make sure to let a healthcare professional know if you have a latex allergy.
Stelara can be injected into any of these sites:
- upper front thigh
- abdomen, except for a 2-inch radius around your belly button
- outer upper arm
Before injecting, always check the expiration date on the box. You should also inspect the liquid in the syringe. It should range in color from clear to pale yellow. It may contain a few small, white particles. If the liquid is cloudy, frozen, dark yellow, or has large particles, do not use it.
Don’t Miss: What To Take For Stomach Ulcer Pain
Before Taking This Medicine
You should not use Stelara if you are allergic to ustekinumab, or if you have:
-
active tuberculosis or
-
if you have received a BCG vaccine within the past 12 months.
Tell your doctor if you have ever had tuberculosis, if anyone in your household has tuberculosis, or if you have recently traveled to an area where tuberculosis is common.
To make sure Stelara is safe for you, tell your doctor if you have:
-
signs of infection
-
chronic infections
-
if you recently received or are scheduled to receive any vaccine.
Some people using Stelara have developed skin cancer . However, these people may have had a higher risk of skin cancer. Talk to your doctor about this risk and what skin symptoms to watch for. You may need to have regular skin exams.
It is not known whether this medicine will harm an unborn baby. Tell your doctor if you are pregnant or plan to become pregnant.
If you are pregnant, your name may be listed on a pregnancy registry to track the effects of ustekinumab on the baby.
It may not be safe to breast-feed while using this medicine. Ask your doctor about any risk.
Us Food And Drug Administration
- Moderate to severe plaque psoriasis in patients 6 years or older who are candidates for phototherapy or systemic therapy
- Active psoriatic arthritis
- Moderately to severely active Crohns disease
- Moderately to severely active ulcerative colitis
Ustekinumab is available as Stelara . Ustekinumab is a human interleukin-12 and -23 antagonist. IL-12 and IL-23 are naturally occurring cytokines that are involved in inflammatory and immune responses, such as natural killer cell activation and CD4+ T-cell differentiation and activation. In in vitro models, ustekinumab was shown to disrupt IL-12 and IL-23 mediated signaling and cytokine cascades by disrupting the interaction of these cytokines with a shared cell-surface receptor chain, IL-12R1. The cytokines IL-12 and IL-23 have been implicated as important contributors to the chronic inflammation that is a hallmark of Crohns disease and ulcerative colitis. In animal models of colitis, genetic absence or antibody blockade of the p40 subunit of IL-12 and IL-23, the target of ustekinumab, was shown to be protective .
The most common adverse reactions include the following:
Don’t Miss: Cure For Peptic Ulcer By Diet
Fda Approves Stelara For Treatment Of Moderate
NEW YORK, NY The Crohns & Colitis Foundation released the following statement today after the U.S. Food & Drug Administration announced its approval of STELARA® , a biologic therapy from Janssen Biotech, Inc., for the treatment of moderately to severely active Crohns disease in adults earlier this week. STELARA will be available for use in adult patients with Crohns disease who have failed or were intolerant to treatment with immunomodulators or corticosteroids but never failed treatment with a tumor necrosis factor blocker. It has also been approved for use in patients who failed or were intolerant to treatment with one or more TNF blockers.
“Because of the individual nature of these diseases, what works for one patient may not work for another. That is why it is so critical that our Crohns patients have many different treatment options available to them, said Michael Osso, President & CEO of the Crohn’s & Colitis Foundation. “The approval of STELARA is extremely important for patients living with moderate-to-severe Crohns disease. Many of these patients have exhausted available treatments, and STELARA gives them another option to hopefully, induce remission, help manage their disease, and improve their quality of life.
STELARA® is the first biologic therapy for Crohns disease targeting Interleukin-12 and Interleukin-23 cytokines, which play a key role in inflammatory and immune responses.
About Crohn’s Disease
About STELARA®
Serious Infections
Cancers
Stelara Forms And Strengths
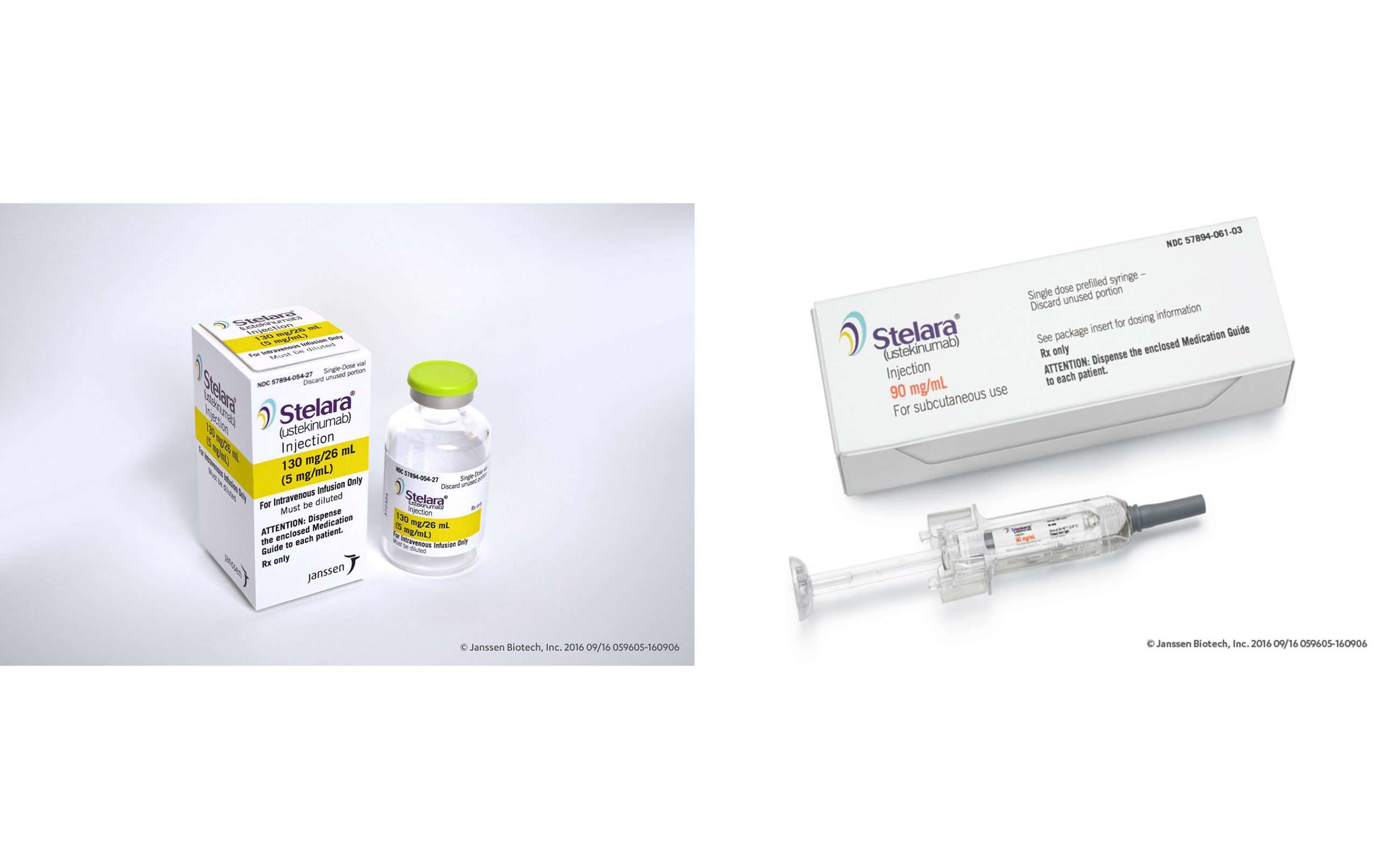
Stelara comes as a liquid solution and contains the active drug ustekinumab. Its available in the following forms and strengths:
- A single-dose prefilled syringe for subcutaneous injection thats available in two different strengths: 45 milligrams per 0.5 milliliters and 90 mg/mL.
- A single-dose vial for subcutaneous injection thats used with a syringe and needle to draw up a dose. This vial is available in one strength: 45 mg/0.5 mL.
- A single-dose vial for intravenous infusion, which is administered only by healthcare professionals. This vial is available in one strength: 5 mg/mL.
Don’t Miss: Symptoms Of Stomach Ulcers In Horses
Posterior Reversible Encephalopathy Syndrome
Two cases of posterior reversible encephalopathy syndrome , also known as Reversible Posterior Leukoencephalopathy Syndrome , were reported in clinical trials. Cases have also been reported in postmarketing experience in patients with psoriasis, psoriatic arthritis and Crohn’s disease. Clinical presentation included headaches, seizures, confusion, visual disturbances, and imaging changes consistent with PRES a few days to several months after ustekinumab initiation. A few cases reported latency of a year or longer. Patients recovered with supportive care following withdrawal of ustekinumab.
Monitor all patients treated with Stelara® for signs and symptoms of PRES. If PRES is suspected, promptly administer appropriate treatment and discontinue Stelara®.
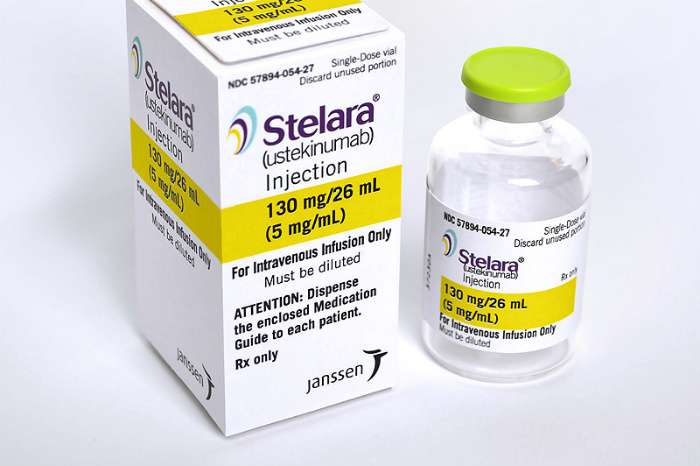
How Supplied/storage And Handling
Stelara® injection is a sterile, preservative-free, colorless to light yellow solution and may contain a few small translucent or white particles. It is supplied as individually packaged, single-dose prefilled syringes or single-dose vials.
For Subcutaneous Use
- 45 mg/0.5 mL
- 90 mg/mL
Each prefilled syringe is equipped with a 27 gauge fixed ½ inch needle, a needle safety guard, and a needle cover that contains dry natural rubber.
Single-dose Vial
- 45 mg/0.5 mL
For Intravenous Infusion
- 130 mg/26 mL
Storage and Stability
Stelara® vials and prefilled syringes must be refrigerated at 2°C to 8°C . Store Stelara® vials upright. Keep the product in the original carton to protect from light until the time of use. Do not freeze. Do not shake.
If needed, individual prefilled syringes may be stored at room temperature up to 30°C for a maximum single period of up to 30 days in the original carton to protect from light. Record the date when the prefilled syringe is first removed from the refrigerator on the carton in the space provided. Once a syringe has been stored at room temperature, it should not be returned to the refrigerator. Discard the syringe if not used within 30 days at room temperature storage. Do not use Stelara® after the expiration date on the carton or on the prefilled syringe.
Don’t Miss: What Foods Should You Eat If You Have An Ulcer
How Effective Are Stelara And Humira
You may wonder whether Stelara and Humira are effective at treating your autoimmune or inflammatory condition.
Both drugs are used to treat certain active forms of plaque psoriasis, psoriatic arthritis, Crohns disease, and ulcerative colitis in adults. Active means you have inflammation and symptoms. See the sections below for information on the drugs effectiveness for these conditions.
To find out the effectiveness of Stelara and Humira for treating conditions in children, talk with your childs doctor. And to learn about the effectiveness of Humira for uses other than those listed below, see this in-depth article.
Stelara Shows Favorable Safety Profile Through 1
Disclosures: We were unable to process your request. Please try again later. If you continue to have this issue please contact .
Data from six phase 2/3 studies showed that Stelara had a favorable safety profile through 1-year of treatment in Crohns disease and ulcerative colitis, according to research published in Inflammatory Bowel Diseases.
William J. Sandborn, MD, of the University of California, San Diego, and colleagues wrote that previous research including in psoriatic disease has demonstrated a good safety profile for the drug , but integrated analysis of combined patients with IBD, could help physicians make treatment choices.
This increases the precision to detect safety signals compared with placebo for all events and increases identification of less frequent events, they wrote. Furthermore, because ustekinumab IBD indications uniquely employ intravenous induction, followed by subcutaneous maintenance dosing these integrated safety analyses of all IBD phase 2/3 studies also provide important data examining the possibility that these higher doses or the IBD population might have a different safety profile than the psoriatic diseases.
Researchers pooled data from six ustekinumab phase 2/3 CD and UC studies and evaluated safety through 1 year. Patients received either placebo or induction and maintenance therapy with ustekinumab .
Also Check: What To Avoid Eating With Ulcerative Colitis
Pharmacoeconomic Review Report: Ustekinumab
Indication: Treatment of adult patients with moderately to severely active ulcerative colitis who have had an inadequate response with, lost response to, or were intolerant to either conventional therapy or a biologic or have medical contraindications to such therapies
CADTH Common Drug Review
Copyright Act
Ustekinumab is indicated for use in adult patients with moderately to severely active ulcerative colitis who have had an inadequate response, or were intolerant to either conventional therapy or a biologic, or have medical contraindications to such therapies. The recommended dose of ustekinumab is a single IV tiered infusion based on body weight during the induction phase, followed by subcutaneous injections of 90 mg every eight weeks during the maintenance phase. The sponsor-submitted price is $2,079.84 per 130 mg/26 mL solution vial for IV infusion and $4,593.14 for a pre-filled syringe of 90 mg/1 mL for subcutaneous injection. The cost of treatment per patient with ustekinumab is estimated to be $33,798 in the first year and increases to $32,152 annually thereafter.
The sponsor submitted a cost-utility analysis comparing ustekinumab with other biologic therapies or continuing conventional therapy for Canadian adults with moderately to severely active UC and an inadequate, intolerant, or failed response to CT or biological drugs.
Takeaway And Helpful Resources
The dosages in this article are typical ones provided by the drugs manufacturer. If your doctor recommends Stelara for you, they will prescribe the dosage thats right for you. Always follow the dosage that your doctor prescribes for you.
As with any drug, never change your dosage of Stelara without your doctors approval. If you have questions about the dosage of Stelara thats right for you, talk with your doctor.
Besides learning about the dosages, you may want other information about Stelara. These additional articles might be helpful to you:
- More about Stelara. For information about other aspects of Stelara, refer to this article.
- Side effects. To learn about side effects of Stelara, see this article. You can also look at the Stelara medication guide.
- Drug comparison. To find out how Stelara compares with other drugs, read the comparison articles about Cosentyx, Humira, and Otezla.
- Details about your condition. For details about your condition, see our:
Recommended Reading: Can You Donate Plasma If You Have Ulcerative Colitis
How Stelara Is Given
Be sure to use Stelara according to your doctors instructions.
Stelara is given by subcutaneous injection for most of the conditions that its approved to treat. Your healthcare professional will give you your first dose. They may also show you how to inject the medication yourself for future doses. The drug can be given under the skin of your abdomen , thighs, buttocks, or upper arms.
If youre using Stelara to treat ulcerative colitis or Crohns disease, youll receive your first dose as an intravenous infusion. This is an injection into a vein given over a period of time. A healthcare professional will give you the IV infusion at a clinic, doctors office, or infusion center. After your first dose, youll receive Stelara as subcutaneous injections.
To help you give yourself Stelara injections, the drugs manufacturer hasstep-by-step instructions. It also provides instructional videos about how to self-inject Stelara.
Its important that you dont use more Stelara than your doctor prescribes. For some medications, taking more than the recommended amount may lead to side effects or overdose.
What Is The Cost Of Stelara And Humira
Whether you have health insurance or not, cost may be a factor when youre considering these drugs. To see cost estimates for Stelara and Humira based on where you live, visit GoodRx.com.
Keep in mind that what youll pay for either drug will depend on your treatment plan, health insurance, and the pharmacy you use. It may also depend on the cost of any visits to your healthcare provider to receive doses of Stelara or Humira.
Stelara and Humira are both brand-name biologic drugs. Biologic drugs dont have generic versions. However, some have biosimilar versions.
Biosimilars are medications that are similar to original brand-name biologics.
Generics are exact copies of brand-name drugs. Biosimilars, on the other hand, have the same active ingredients as the parent biologic but arent exactly the same. However, biosimilars are considered to be as safe and effective as brand-name biologics.
Like generics, biosimilars usually cost less than their brand-name parent drugs.
Stelara and Humira dont yet have biosimilars available.
Get answers to some frequently asked questions about Stelara and Humira.
You May Like: Vsl 3 For Ulcerative Colitis
Dosage Forms And Strengths
Stelara® is a colorless to light yellow solution and may contain a few small translucent or white particles.
Subcutaneous Injection
- Injection: 45 mg/0.5 mL or 90 mg/mL solution in a single-dose prefilled syringe
- Injection: 45 mg/0.5 mL solution in a single-dose vial
Intravenous Infusion
- Injection: 130 mg/26 mL solution in a single-dose vial
What Is Crohns Disease
Crohns disease is a type of inflammatory bowel disease. The cause of Crohns is currently unknown, although genetics, heredity, and environmental factors may all play a role.
People with this condition have chronic inflammation in their digestive tract. This is caused by an exaggerated immune system response to the beneficial bacteria that live in the gut.
Crohns affects people differently. Some symptoms you may experience with Crohns include:
- diarrhea
Read Also: I Think I Have An Ulcer What Should I Do
Theoretical Risk For Vulnerability To Particular Infections
Individuals genetically deficient in IL-12/IL-23 are particularly vulnerable to disseminated infections from mycobacteria , salmonella , and Bacillus Calmette-Guerin vaccinations. Serious infections and fatal outcomes have been reported in such patients.
It is not known whether patients with pharmacologic blockade of IL-12/IL-23 from treatment with Stelara® may be susceptible to these types of infections. Appropriate diagnostic testing should be considered, e.g., tissue culture, stool culture, as dictated by clinical circumstances.
Ustekinumab Gains Fda Approval As Ulcerative Colitis Treatment
Fourth indication expansion for the 2009 drug.
The FDA has granted the approval of an indication expansion for Johnson & Johnsons ustekinumab for the treatment of adults with moderately to severely active ulcerative colitis.
Ustekinuamb is a human interleukin-12 and -23 antagonist originally approved in 2009 for the management of moderate to severe plaque psoriasis in adult patients. From there, the FDA approved three more indication expansions, including the uses to treat adolescent patients with moderate to severe plaque psoriasis, adults with active psoriatic arthritis, and adults with moderately to severely active Crohns disease.
Because of the individual nature of ulcerative colitis, what works for one patient may not work for another. That is why it is so critical that our ulcerative colitis patients have many different treatment options available to them, said Caren Heller, MD, Chief Scientific Officer at the Crohns & Colitis Foundation in a statement. The approval of STELARA is extremely important for patients living with moderate to severe ulcerative colitis. STELARA gives patients another option to, hopefully, induce remission and help manage their disease.
Trending: Top 10 Best States to Be a Pharmacist in 2019
In the induction arm, 19% of patients achieved clinical remission by week eight, whereas 58% experienced some form of a clinical response by week eight.
Recommended Reading: What To Take For An Ulcer Over The Counter