Researchers Engineer Novel Disease Model To Develop New Ulcerative Colitis Drugs
Dr. Huang developed an innovative patient-derived model of the chronic inflammatory bowel disease that mirrors the diseases complexity better than others currently available and identified the protein CXCL8 as an actionable drug target.
01/11/2021
As reported in Nature Communications, researchers from the Department of Cancer Biology have developed a novel, patient-derived model of ulcerative colitis, which will help advance studies into new treatments for the chronic inflammatory bowel disease. The team used the model to identify a promising target that could be inhibited to slow disease progression.
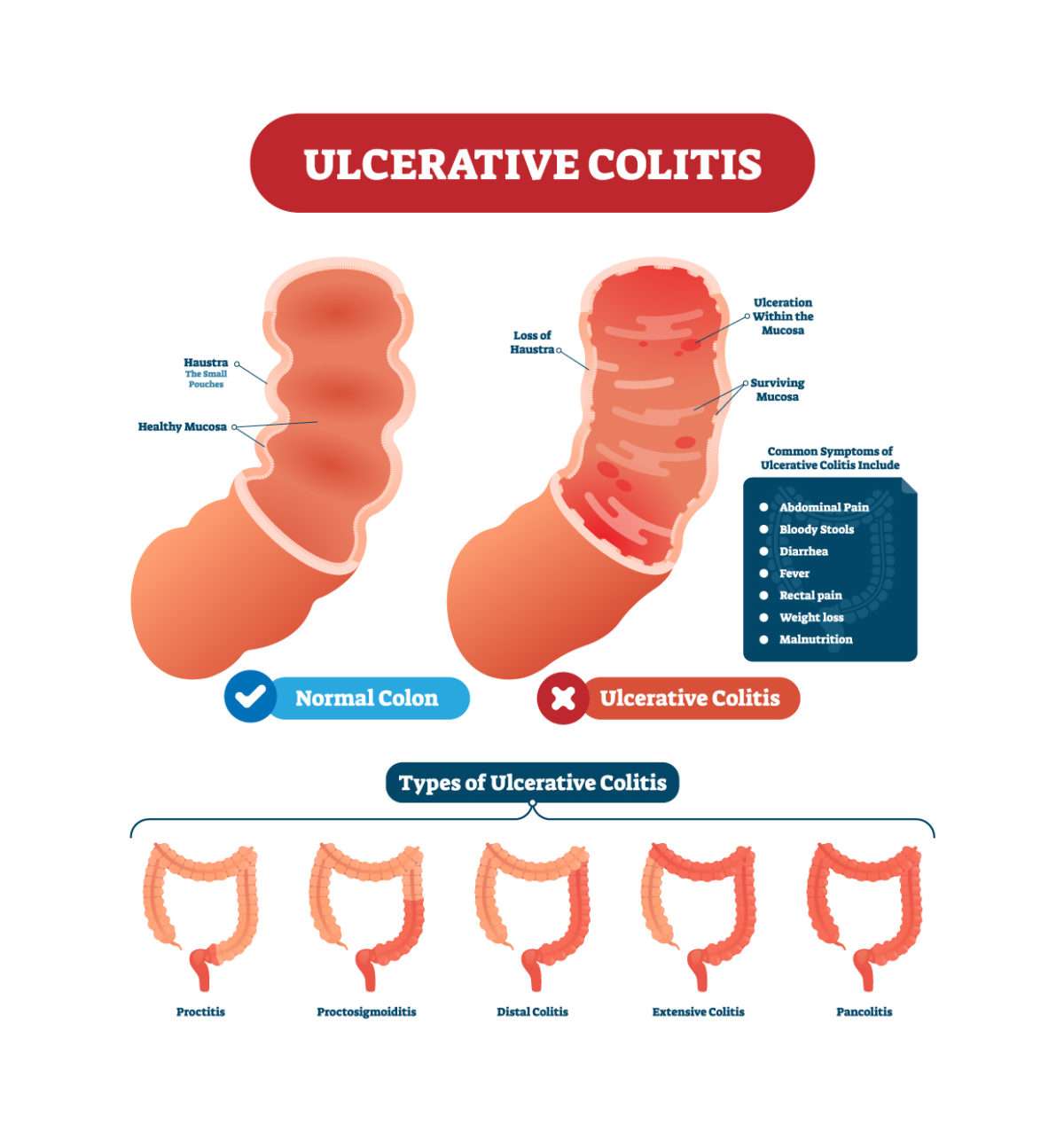
Ulcerative colitis is characterized by abnormal reactions of the immune system that lead to inflammation and ulcers on the inner lining of the large intestines. It is a highly heterogeneous condition, both in terms of patients symptoms and disease pathology.
Suppressing the overactive immune response with anti-inflammatory drugs is the current mainstay treatment for inflammatory bowel diseases, although these therapies have limited long-term efficacy in ulcerative colitis patients.
Recent Advances And New Biomarkers In Ulcerative Colitis
Ulcerative colitis is a chronic inflammatory bowel disease characterized by the occurrence of many pathological lesions, such as ulcers, crypt abscesses, small vessel inflammation leading to inflammation, and ulcerative lesions of the mucosa and submucosa, that are the main pathological features of the …
Keywords:ulcerative colitis, biomarkers, therapies, artificial intelligence, machine learning, mucosal healing
Important Note: All contributions to this Research Topic must be within the scope of the section and journal to which they are submitted, as defined in their mission statements. Frontiers reserves the right to guide an out-of-scope manuscript to a more suitable section or journal at any stage of peer review.
Cholesterol Sulfate Alleviates Ulcerative Colitis By Promoting Cholesterol Biosynthesis In Colonic Epithelial Cells
New treatment strategies are required for ulcerative colitis. Here the authors show in mouse models that cholesterol sulfate, an endogenous active cholesterol derivative, contributes to the healing of the mucosal barrier by promoting cholesterol biosynthesis in colonic epithelial cells and exhibits therapeutic efficacy against ulcerative colitis.
17 June 2022 | Open Access
Also Check: What Are The Symptoms Of Stomach Ulcers In Humans
Predicting The Best Treatment For Young Ulcerative Colitis Patients
Children and teens with ulcerative colitis have many more treatment options than a decade ago, thanks to the invention of several new medications for this form of inflammatory bowel disease. But not all drugs work for every patient, meaning it can take weeks or months of trial and error to bring someones symptoms under control.
Pediatric gastroenterologistMichael Rosen, MD, director of the Center for Pediatric Inflammatory Bowel Disease and Celiac Disease at Stanford Medicine Childrens Health, works with a team to care for young patients with Crohns disease, ulcerative colitis and celiac disease, all of which can cause inflammation in the gut. They also conduct research to understand the biology of these conditions, devise better treatments and advance precision medicine for these diseases.
People with ulcerative colitis experience unpleasant symptoms, such as intestinal pain, frequent bloody diarrhea, weight loss, anemia and fatigue. Multiple parts of the bowel, including the epithelial barrierthe cells that line the bowelare impaired, and the immune response becomes overzealous, likely more in response to changes in the bacteria populating the colon than to patients own cells. The bowel lining becomes inflamed and ulcerated, leading to blood loss.
Dr. Michael Rosen and his team are studying how to match patients to the best-for-them ulcerative colitis treatments using gene signals.
Why was your new study important?
Clinical Trials For Uc
Clinical trials are studies that use people to test new forms of treatment. A person can sign up for a clinical trial as long as they meet the criteria for the study. Before signing up, they should talk with a doctor about the possible risks and benefits of joining an investigational study.
People who are interested in joining a study can also talk with a doctor to see if they know of any upcoming studies they may qualify for. Interested people can also visit ClinicalTrials.gov to find UC studies across the country.
You May Like: How Do You Treat Diabetic Ulcers
Navigating The Road To Prevention
The physicians at the Susan and Leonard Feinstein IBD Center are focusing their work on helping patients suffering with IBD. We create customized treatment plans based on the latest research spearheaded by members of our research team. We are continuously studying ways to predict who will develop symptoms and make the most of therapies before symptoms start.
Our research team has also developed a very exciting program, Life Before IBD, dedicated to studying aspects of each patients life before developing IBD. The overall goal of this project is to define important developments in the gastrointestinal tract before IBD occurs. We plan to study risks for IBD in large families with many members who have IBD. We also want to study babies of mothers with IBD and siblings of IBD patients.
We believe that the results of Life Before IBD study may help us understand how we can prevent the disease from ever happening, especially in high-risk groups.
To find out more about our Life Before IBD program, or if youre eligible to participate because you have a large family with IBD, please email us at .
The Upadacitinib Study Coordinated By San Raffaele
The study – multicentre, randomized, double-blind and placebo-controlled – consisted of three “sub-studies” with different aims: two induction studies, in which the effect of upadacitinib was evaluated compared to placebo in the initial phase of the treatment – when we need to turn off the ongoing inflammation – and a maintenance study, to observe the effects in the medium to long term.
The clinical trial was conducted in Europe, North and South America, Australia, Africa and some Asian regions. The enrolled patients were aged between 16 and 75 years, have been suffering from moderate to severe ulcerative colitis for at least 90 days and had inadequate responses to conventional therapies or medical contraindications to undergo such treatments. Explains Prof. Silvio Danese:
During the induction studies, we treated nearly 1000 patients with 45 mg of upadacitinib or placebo once a day for 8 weeks, with excellent clinical, endoscopic and histological efficacy results. Those who showed good clinical remission from the drug – 451 patients in all – were randomly re-assigned to upadacitinib at different doses or to placebo for the maintenance phase of the study, which lasted another 52 weeks.
The results show that the new drug is safe, effective and well tolerated both in the short and medium to long term, making it an excellent therapeutic option for patients suffering from ulcerative colitis even compared to other drugs already on the market.
Recommended Reading: Rice Recipes For Ulcerative Colitis
Stanford Scientists Link Ulcerative Colitis To Missing Gut Microbes
Bacteria normally inhabiting healthy peoples intestines and the anti-inflammatory metabolites these bacteria produce are depleted in ulcerative colitis patients, a Stanford study shows.
Aida Habtezion is the senior author of a study that describes how people with ulcerative colitis have insufficient amounts of a metabolite produced by a family of gut-dwelling bacteria.Steve Castillo
About 1 million people in the United States have ulcerative colitis, a serious disease of the colon that has no cure and whose cause is obscure. Now, a study by Stanford University School of Medicine investigators has tied the condition to a missing microbe.
The microbe makes metabolites that help keep the gut healthy.
This study helps us to better understand the disease, said Aida Habtezion, MD, associate professor of gastroenterology and hepatology. We hope it also leads to our being able to treat it with a naturally produced metabolite thats already present in high amounts in a healthy gut.
When the researchers compared two groups of patients one group with ulcerative colitis, the other group with a rare noninflammatory condition who had undergone an identical corrective surgical procedure, they discovered that a particular family of bacteria was depleted in patients with ulcerative colitis. These patients also were deficient in a set of anti-inflammatory substances that the bacteria make, the scientists report.
What Is Upadacitinib And How It Works
Upadacitinib is an oral drug already used for the treatment of certain diseases, such as rheumatoid arthritis, psoriatic arthritis, ankylosing spondylitis and atopic dermatitis. In particular, its task is to block specific pro-inflammatory cytokines involved in the intestinal inflammation process and highly present in the tissues of patients with ulcerative colitis.
Unlike other molecules, upadacitinib is able to inhibit several cytokines at the same time and thus to better control different pathways of inflammation. That’s why the researchers thought that its use could also be effective in patients with ulcerative colitis.
Read Also: Best Treatment For Diabetic Foot Ulcer
Safety Of Studies And Clinical Trials
Clinical trials and other research featured and/or supported by the Crohns & Colitis Foundation are conducted under thorough oversight by an ethics committee along with scientists and clinical advisors to ensure the safety of all participants. All research, however, carries some form of risk.
Heres what we do to ensure the safety of everyone who participates in one of our research initiatives:
-
All research opportunities listed by the Foundation are required to provide official documentation that the trial has been approved by the Institutional Review Board of the participating research facility.
-
Each research listing is reviewed by the Crohns & Colitis Foundation’s National Scientific Advisory Committee.
-
Except for studies funded by the Crohns & Colitis Foundation, we do not endorse or ensure the scientific merit of the trials we list. If you are considering participating in a study, you should discuss the opportunity with your physician.
Good Bacteria Show Promise For Clinical Treatment Of Crohns Disease Ulcerative Colitis
May 28, 2021
Balfour Sartor, MD, Midget Distinguished Professor of Medicine, Microbiology and Immunology, is the senior author of a study that shows how a novel consortium of bacteria that live in the digestive tracts of healthy individuals can be used to prevent and treat aggressive colitis in humanized mouse models.
CHAPEL HILL, NC A new study published in Nature Communications demonstrates that a consortium of bacteria designed to complement missing or underrepresented functions in the imbalanced microbiome of inflammatory bowel disease patients, prevented and treated chronic immune-mediated colitis in humanized mouse models. The studys senior author, Balfour Sartor, MD, Midget Distinguished Professor of Medicine, Microbiology and Immunology, Co-Director of the UNC Multidisciplinary IBD Center, said the results are encouraging for future use treating Crohns disease and ulcerative colitis patients.
The idea with this treatment is to restore the normal function of the protective bacteria in the gut, targeting the source of IBD, instead of treating its symptoms with traditional immunosuppressants that can cause side effects like infections or tumors, Sartor said.
It also decreased pathobionts bacteria that can cause harm while expanding resident protective bacteria, and produced metabolites promoting mucosal healing and immunoregulatory responses, Sartor said. Simply put the treatment increased the good guys and decreased the bad guys.
Don’t Miss: What Can I Drink With Ulcerative Colitis
Ulcerative Colitis: Bacteria Findings Raise Hopes For New Treatment
Sufferers found to have low levels of gut microbes that convert bile acids into other substances
Hopes of developing a new treatment for ulcerative colitis have been raised by research suggesting the condition may be linked to low levels of certain bacteria in the gut.
Ulcerative colitis is a long-term condition in which the colon becomes inflamed. According to the NHS, it is thought to be an autoimmune disease and affects one in every 420 people in the UK. Sufferers need to empty their bowels frequently, develop ulcers in their colon and have recurring bouts of diarrhoea with blood and pus in the faeces.
While a number of medicines can help manage the condition, ulcerative colitis can only be cured by removing the colon. However, about half of those who have this surgery go on to have inflammation in the area of the small intestine that is reconstructed into a pouch to serve as a rectum. Such inflammation is treated with antibiotics.
Now researchers say patients who have had this surgery for ulcerative colitis have lower levels of certain acids in their faeces, and fewer of the bacteria that produce them, than those who have had the surgery for other reasons.
The team say the results shed light on why the latter have a far lower risk of inflammation in their pouch. They say it also offers insights into ulcerative colitis itself, with experiments in mice suggesting inflammatory bowel diseases could be treated by introducing these missing acids into the colon.
Questions To Consider Before Signing Up
Participating in a clinical trial may not be the best choice for everyone. A person should bear a range of factors in mind before signing up. A doctor may be able to help answer some of their questions.
Individuals who are interested in signing up for a clinical trial should consider the following questions first:
- What are the costs associated with the trial?
- How safe is the trial?
- Is additional follow-up care available after the trial?
- What is the desired personal outcome from participation?
- Who is eligible to participate?
- What is the main purpose of the trial?
- What are the researchers expecting of the person?
- What effects will the trial have on the persons regular healthcare routines?
A person can find clinical trials online. In some cases, they may not be able to participate due to the location of the study.
Two websites that a person can check out to find clinical studies are:
Crohns & Colitis Foundation Research Finder
This free tool, which provides several filters to help narrow down the search, can help a person find UC studies around the United States.
ClinicalTrials.gov
People can also use this free tool to look for studies on UC or any other health condition. A person can type in the condition only or add additional filters to narrow down the results.
Don’t Miss: Icd 10 Stage 4 Sacral Ulcer
What Have We Learned About Ulcerative Colitis From Niddk
The NIDDK has supported many studies to learn more about ulcerative colitis and other types of IBD. NIDDK-supported research efforts include
- the IBD Genetics Consortium , established in 2002 to identify genes that make some people more likely to develop IBD. The IBDGC, in collaboration with the International IBD Genetics Consortium, has enrolled thousands of people with IBD and identified about 200 regions of the human genome that are associated with risk of IBD.
- the Predicting Response to Standardized Pediatric Colitis Therapy study, which has identified genetic factors and patient characteristics that may help researchers develop more personalized and effective treatment approaches for children with ulcerative colitis.
What Are Clinical Trials For Ulcerative Colitis
Clinical trialsand other types of clinical studiesare part of medical research and involve people like you. When you volunteer to take part in a clinical study, you help doctors and researchers learn more about disease and improve health care for people in the future.
Researchers are studying many aspects of ulcerative colitis and other types of inflammatory bowel disease , such as
- genes that are associated with IBD in African Americans
- whether a special diet can alter the bacteria in the large intestine and induce remission in children with ulcerative colitis
- how the immune system controls inflammation in the digestive tract in people with IBD
- new treatments for ulcerative colitis
Watch a video of NIDDK Director Dr. Griffin P. Rodgers explaining the importance of participating in clinical trials.
Read Also: Can You Eat Cheese With Ulcerative Colitis
Once Daily Dosing Of Delayed
In the QDIEM trial, Sandborn and colleagues investigated the use of once-daily dosing of delayed-release mesalamine compared with divided dosing in UC patients.7 The study was designed to assess the non-inferiority of once-daily versus twice-daily dosing for maintaining clinical remission. Patients who had been maintained on mesalamine doses ranging from 1.6 g/d to 2.4 g/d were randomized to receive either once-or twice-daily regimen of the same overall dose they had been receiving before the study began. Treatments continued for 12 months, with patient visits at 3, 6, and 12 months and a phone call at 9 months. The primary endpoint was the percentage of patients remaining in remission at month 6 for each dosing arm. Relapse was defined as a Simple Clinical Colitis Activity Index score of 5 or higher, and remission was defined as a SCCAI score of 2 or less.
Ulcerative Colitis Medication And Treatments
Ulcerative colitis treatment consists of a variety of approaches. Most patients can control their symptoms with ulcerative colitis drugs. In general, ulcerative colitis surgery is reserved for the most severe cases and those in which standard ulcerative colitis medication is ineffective.
5-aminosalicylic acid drugs are the most common ulcerative colitis medications. Corticosteroids are used for short-term treatment of moderate symptoms. Severe cases receive immunomodulators, which reduce inflammation but slow the bodys immune response.
Surgery can be used to remove damaged areas of the digestive tract. In profound cases, ulcerative colitis may be eliminated by removing the colon and rectum completely, a procedure called proctocolectomy.
Recommended Reading: Can Ulcers Cause Bloody Stool
Rapid Symptom Resolution With Delayed
Sandborn and associates also provided a retrospective, post-hoc analysis of data from ASCEND I4 and ASCEND II 5 trials. In this analysis, the researchers sought to evaluate the time to resolution of hallmark UC symptoms in 423 patients with moderately active UC with a history of difficult-to-treat disease.8 Hallmark symptoms were defined as increased stool frequency and rectal bleeding, and moderately active disease was defined as a score of 2 on the Physician’s Global Assessment. For this analysis, difficult-to-treat disease was defined as that previously treated with UC therapies .
The investigators analyzed data from ASCEND I and II and compared the median time to resolution for delayed-release mesalamine 4.8 g/day compared with 2.4 g/day dosing. The time to resolution was based on the first day of symptom resolution as reported by patients, and resolution was defined as a score of 0 on the Physician’s Global Assessment. The mean age of patients randomized to receive 2.4 g/day and 4.8 g/day was 42.6 and 43.4, respectively. The mean UC Disease Activity Index score was 7.3 at the beginning of the study for both study arms.