Bristol Myers Squibb Presents Interim Results From Long
The percentage of patients achieving clinical remission, clinical response, endoscopic improvement and corticosteroid-free remission was maintained through Week 142
Zeposia is the first and only oral sphingosine 1-phosphate receptor modulator approved to treat patients with ulcerative colitis
PRINCETON, N.J.â-Bristol Myers Squibb today announced interim results from the True North open-label extension study evaluating the long-term efficacy and safety profile of Zeposia in patients with moderately to severely active ulcerative colitis . Findings show that the percentage of patients achieving clinical remission, clinical response, endoscopic improvement and corticosteroid-free remission was maintained through Week 142. No new safety signals emerged in the study. These data will be presented at the 17th Congress of the European Crohnâs and Colitis Organisation , taking place February 16-19, 2022.
Additional Bristol Myers Squibb-sponsored abstracts presented at the ECCO 2022 Congress can be accessed online here.
Visit this page on BMS.com for more information on Bristol Myers Squibbs scientific approach and resources on gastrointestinal immune-mediated diseases.
About True North
Bristol Myers Squibb thanks the patients and investigators involved in the True North clinical trial.
About Ulcerative Colitis
ZEPOSIA is indicated for the treatment of:
IMPORTANT SAFETY INFORMATION
About Bristol Myers Squibb
E Targeting Leukocyte Circulation Outside The Intestine
Blockade of lymphocyte recirculation in lymph nodes can be targeted through modulation of sphingosine 1-phosphate receptors. Sphingosine 1-phosphate is a bioactive lysophospholipid metabolite that can act as an intercellular lipid mediator of inflammation. Interaction between S1P and the five known S1P receptors regulate the response and function of biological processes, including cell differentiation, migration, proliferation and trafficking of T and B cells.
Fingolimod was the first S1P receptor modulator to reach the market, initially approved for the treatment of MS . The drug is phosphorylated in vivo, acquiring a structure similar to that of S1P, and functions as a non-selective small-molecule agonist to four of the five S1P receptors. Fingolimod causes peripheral blood lymphopenia due to sequestration of lymphocytes within lymphoid tissue and is associated with cardiovascular side effects, such as bradycardia and hypotension. This led to the development of more specific S1P receptor modulators with preferential actions on S1PR1, S1PR4 and S1PR5, since fingolimod also showed beneficial effects in murine models of colitis .
Carolyn Goldschmidt DO, Marisa P. McGinley DO, MSc, in, 2021
Ozanimod As Therapy For Ulcerative Colitis
- The New England Journal of Medicine
You’ve saved your first item
You can find your saved items on your dashboard, in the “saved” tab.
You’ve recommended your first item
Your recommendations help us improve our content suggestions for you and other PracticeUpdate members.
You’ve subscribed to your first topic alert
What does that mean?
Don’t Miss: How Do You Get Pressure Ulcers
The Effects Of Maintenance Therapy With Upadacitinib On Abdominal Pain Bowel Urgency And Fatigue In Patients With Moderately To Severely Active Ulcerative Colitis: Phase 3 U
Upadacitinib is a reversible, selective Janus kinase inhibitor.1 In the phase 3 U-ACHIEVE and U-ACCOMPLISH trials, induction therapy with upadacitinib was superior to placebo in patients with moderately to severely active ulcerative colitis who required treatment after previous therapy.2,3 Improvements were reported in symptoms such as abdominal pain, bowel urgency, and fatigue, which can be debilitating to these patients.4
Patients who demonstrated a clinical response during the 8-week induction period with daily upadacitinib were enrolled in the U-ACHIEVE maintenance trial. Silvio Danese, MD, PhD, presented results for this cohort.5This study randomly assigned 451 patients to receive upadacitinib at 15 mg, upadacitinib at 30 mg, or placebo, in a double-blind manner. Patient-reported outcomes of abdominal pain and bowel urgency were assessed during maintenance treatment. The Functional Assessment of Chronic Illness Therapy Fatigue instrument was used to measure fatigue. A change of 5 or more points from baseline in the FACIT-F score was considered a meaningful within-person change, and an increase of 40 or more points was considered normalization of fatigue.
References
1. Kim JW, Kim SY. The era of Janus kinase inhibitors for inflammatory bowel disease treatment. Int J Mol Sci. 2021 22 :11322.
2. Sandborn WJ, Ghosh S, Panes J, et al. Efficacy of upadacitinib in a randomized trial of patients with active ulcerative colitis. Gastroenterology. 2020 158:2139-2149.e14.
Efficacy And Safety Of Combination Induction Therapy With Guselkumab And Golimumab In Participants With Moderately To Severely Active Ulcerative Colitis: Results Through Week 12 Of A Phase 2a Randomized Double
Guselkumab is an antagonist of the p19 subunit of IL-23 that is approved to treat plaque psoriasis. Golimumab is an antagonist of TNF and is approved for the treatment of ulcerative colitis.1 The phase 2a VEGA study evaluated the safety and efficacy of induction therapy with guselkumab plus golimumab vs monotherapy with guselkumab or golimumab in adults with moderately to severely active ulcerative colitis.2The trial enrolled patients with a Mayo score of 6 to 12 and an endoscopy subscore of 2 or lower according to central review. The patients had received prior therapy that was either intolerable or unsuccessful. The trial excluded patients previously treated with a TNF antagonist.
Patients were randomly assigned into the 3 arms. In the golimumab monotherapy arm, this agent was administered at 200 mg subcutaneously at week 0, followed by 100 mg administered at weeks 2, 6, and 10. In the guselkumab monotherapy arm, treatment was administered at 200 mg intravenously at weeks 0, 4, and 8. For combination therapy, the 2 antibodies were administered in combination at the same doses and schedules. The induction phase for all 3 arms continued for 12 weeks. The primary endpoint was clinical response, defined as a decrease from baseline in the Mayo score of at least 30% and 3 points, with either a decrease in the rectal bleeding subscore of 1 or more or a rectal bleeding subscore of 0 or 1.
References
You May Like: What Is Acute Ulcerative Colitis
Highlights In Ulcerative Colitis From The 17th Congress Of The European Crohns And Colitis Organisation: Commentary
The 17th Congress of the European Crohns and Colitis Organisation had been scheduled to take place in Vienna, Austria, but was reorganized as a virtual conference based on ongoing concerns about the COVID-19 pandemic. Many advances in the field were shared at the meeting. Here I discuss some of the clinically relevant abstracts about ulcerative colitis, which provided data for treatments such as ozanimod, Janus kinase inhibitors, and selective interleukin 23 inhibitors.
Ozanimod
Ozanimod is the newest available therapy for ulcerative colitis. Ozanimod is a first-in-class sphingosine 1-phosphate receptor modulator that is approved by the US Food and Drug Administration for the treatment of moderately to severely active ulcerative colitis in adults.1 This mechanism is unique in the field of inflammatory bowel disease. Ozanimod is an oral small molecule that targets the signaling molecule S1P, and thereby blocks activated lymphocytes from trafficking through the lymphatic system to reach the inflamed bowel. S1P receptor modulators are also used to treat multiple sclerosis, and ozanimod is approved in this setting. At the 17th Congress of ECCO, several presentations provided efficacy and safety analyses of the pivotal phase 3 True North study of ozanimod.2
JAK Inhibitors
Selective IL-23 Inhibitors
Disclosure
References
4. Kinouchi Y, Hiwatashi N, Chida M, et al. Telomere shortening in the colonic mucosa of patients with ulcerative colitis. J Gastroenterol. 1998 33:343-348.
To Evaluate Efficacy And Long
| The safety and scientific validity of this study is the responsibility of the study sponsor and investigators. Listing a study does not mean it has been evaluated by the U.S. Federal Government.Know the risks and potential benefits of clinical studies and talk to your health care provider before participating. Read our disclaimer for details. |
| First Posted : April 16, 2019Last Update Posted : February 8, 2022 |
| Study Type : | |
| Treatment | |
| Official Title: | A Phase 2/3, Multicenter, Randomized, Double-blind, Placebo-controlled Study of Oral Ozanimod to Evaluate Efficacy and Long-term Safety in Japanese Subjects With Moderately to Severely Active Ulcerative Colitis |
| Actual Study Start Date : |
Read Also: Why Is Ulcerative Colitis Worse At Night
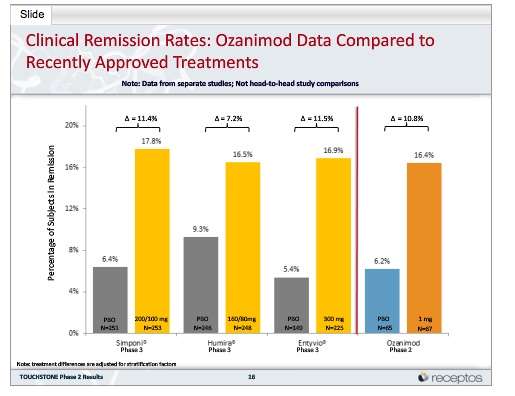
Ozanimod Is Efficacious In Ulcerative Colitis
A phase III, multicentre, randomized, double-blind, placebo-controlled trial investigated ozanimod in patients with moderately to severely active ulcerative colitis. In the 10-week induction period, 645 patients were included in cohort 1 and 367 patients in cohort 2 . In the maintenance period, 457 patients from either cohort with a clinical response to ozanimod were randomly assigned to receive double-blind ozanimod or placebo through to week 52. Clinical remission was significantly higher in the ozanimod group than placebo in both induction and maintenance . The incidence of clinical response was also significantly higher with ozanimod than placebo in both induction and maintenance phases. The overall incidence of adverse events was similar between groups in the induction phase, but higher in the ozanimod group in the maintenance period.
This is a preview of subscription content
Pk Of Ozanimod With Gemfibrozil Itraconazole Or Rifampin
This phase 1, randomized, open-label study focused on assessing the single-dose pharmacokinetics of ozanimod and its metabolites as well as to assess the effects of gemfibrozil, itraconazole, and rifampin on the single-dose PK of ozanimod. A total of 40 patients were randomized to receive either a single oral dose of ozanimod, oral doses of gemfibrozil + a single dose of ozanimod, oral doses of itraconazole + a single dose of ozanimod, or oral doses of rifampin + a single dose of ozanimod. In the single dose of ozanimod alone group, there were dose-proportional increases in Cmax and AUC for both the parent drug, ozanimod as well as its metabolites CC112273 and CC1084037. Itraconazole, a strong inhibitor of CYP3A and P-glycoprotein increased ozanimod AUC by 13%, while rifampin, a strong inducer of CYP3A and P-gp, reduced the AUC of ozanimod by 24%. This implies that there is a CYP3A4 and P-gp involvement in the metabolism of ozanimod. Gemfibrozil, a strong inhibitor of the CYP450 system, increased the AUC for the metabolites of ozanimod, CC112273 and CC1084037 by 47% and 69%, respectively. The metabolites of ozanimod were found to have similar single-dose PK properties, with CYP2C8 being the main enzyme in the metabolism of these metabolites, and CYP3A4 and P-gp being enzymes for the metabolism of ozanimod .
L.R. Fitzpatrick, T. Woldemariam, in, 2017
Don’t Miss: Does Ulcer Pain Come And Go
Efficacy And Safety Of Extended Induction Treatment With Upadacitinib 45 Mg Once Daily Followed By Maintenance Upadacitinib 15 Or 30 Mg Once Daily In Patients With Moderately To Severely Active Ulcerative Colitis
Phase 2b and phase 3 studies have demonstrated the safety and efficacy of upadacitinib when administered daily for 8 weeks as induction treatment for patients with moderately to severely active ulcerative colitis.1-3 A study evaluated the safety and efficacy of 16 weeks of induction therapy with daily upadacitinib at 45 mg, followed by 52 weeks of maintenance therapy with daily upadacitinib administered at 15 mg or 30 mg.4 The patient population consisted of 125 patients with ulcerative colitis without a clinical response after 8 weeks of induction therapy with upadacitinib in the U-ACHIEVE study. Clinical response was defined as a decrease in the adapted Mayo score of 2 or more points and 30% from baseline, plus a decrease of at least 1 point in the rectal bleeding score or an absolute rectal bleeding score of 1 or lower. A response to extended induction therapy was reported in 73 of 125 patients , and 45 of these patients completed the U-ACHIEVE maintenance study.
The rates of adverse events were similar for both doses of maintenance upadacitinib. Similar rates were reported for AEs of special interest, such as anemia , elevated creatine phosphokinase , and serious infection . AEs of special interest that were observed only among the patients who received the higher dose of upadacitinib included hepatic disorder , herpes zoster , neutropenia , adjudicated major adverse cardiovascular events , and nonmelanoma skin cancer .
References
Ozanimod Maintenance In Patients With Ulcerative Colitis Associated With Corticosteroid
|
The following article is a part of conference coverage from the Digestive Disease Week 2021 Annual Meeting , held virtually from May 21 to 23, 2021. The team at Gastroenterology Advisor will be reporting on the latest news and research conducted by leading experts in gastroenterology. Check back for more from DDW 2021. |
For patients with ulcerative colitis, an increased likelihood of achieving corticosteroid-free remission is associated with maintenance treatment with ozanimod vs placebo. This was reported in data presented at the Digestive Disease Week 2021 annual meeting.
Investigators performed further analysis of patients in the maintenance phase of the True North phase 3 trial. Patients with a clinical response to ozanimod at week 10 of the induction phase were re-randomized to either maintenance with 1 mg of ozanimod or placebo. Corticosteroid doses were kept stable during induction then tapered as tolerated during maintenance.
Roughly 33% of patients were on concomitant systemic corticosteroids at baseline. By week 10, 18.4% and 6.0% of the ozanimod and placebo groups achieved clinical remission while on stable corticosteroids, respectively . In the re-randomized maintenance population, 37.0% of the 230 patients in the ozanimod group and 18.5% of the 227 patients in the placebo group achieved clinical remission at week 52 . There was also a higher proportion of patients in corticosteroid-free remission in the ozanimod group vs placebo at week 52 .
Reference
Read Also: Venous Stasis Ulcer Right Lower Leg Icd 10
Ozanimod Yields Clinical Response Remission In Ulcerative Colitis Patients
Serious infections occurred in less than 2% of patients treated with the medication during the duration of the 52-week trial.
Results from both an induction and maintenance therapy trial show ozanimod can be an effective treatment for patients with inflammatory bowel disease .
A team, led by William J. Sandborn, MD, University of California San Diego, compared ozanimod with placebo in achieving both clinical remission and clinical response in patients with moderate to severely active ulcerative colitis.
Ozanimod is a selective sphingosine-1-phosphate receptor modulator currently being studied for the treatment of patients with IBD.
Dont Miss: How To Treat Diabetic Ulcers On Toes
Q: What Are The Limitations Of Current Therapies For Ulcerative Colitis
A: Conventional therapies such as aminosalicylates are modestly effective in patients with moderate, but not severe, disease. Glucocorticoids have been associated with adverse events and long-term adverse health consequences and are not recommended as maintenance therapy. Newer agents, including biologic drugs and Janus kinase inhibitors, are not effective in all patients or can lose efficacy with long-term use, and they have been associated with infections, infusion reactions, and cancers. Thus, the need remains for new oral treatments for ulcerative colitis that are safe and glucocorticoid-sparing and that have durable efficacy.
Read Also: What Over The Counter Medicine For Ulcers
The Efficacy And Safety Of Guselkumab Induction Therapy In Patients With Moderately To Severely Active Ulcerative Colitis: Phase 2b Quasar Study Results Through Week 12
The randomized, double-blind phase 2b QUASAR Induction Study 1 evaluated the safety and efficacy of 12 weeks of guselkumab in patients with moderately to severely active ulcerative colitis.1 The trial enrolled patients previously treated with conventional or advanced therapy that was intolerable or inadequate. Their Mayo rectal bleeding score was 1 or higher at baseline and their Mayo endoscopy subscore was at least 2, based on central review. The patients were randomly assigned to receive placebo, guselkumab at 200 mg every 4 weeks, or guselkumab at 400 mg every 4 weeks. The primary endpoint was the clinical response at week 12.
Among the entire study population of 313 patients, the median age was 41.6±14.40 years, and 59.1% were male. The mean duration of ulcerative colitis was 7.55±6.79 years. The mean Mayo score was 9.2±1.32, and the mean modified Mayo score was 7.0±1.0. Seventy percent of patients had a modified Mayo score of 7, 8, or 9, and 70% of patients had an endoscopy subscore of 3, indicating severe disease. Medications in use at baseline included oral aminosalicylates , oral corticosteroids , and immunosuppressants , and 23.3% of patients were intolerant to 2 or more classes of advanced therapy.
Reference
1. Dignass A, Rubin DT, Bressler B, et al. The efficacy and safety of guselkumab induction therapy in patients with moderately to severely active ulcerative colitis: phase 2b QUASAR study results through week 12 . J Crohns Colitis. 2022 16.
Patient Disposition And Baseline Characteristics
Of 197 patients in TOUCHSTONE, 170 entered the OLE period and received daily ozanimod HCl 1 mg . Of these, 81 patients entered the OLE period at the end of the induction period, seven entered during the maintenance period, and 82 entered at the end of the maintenance period . At the time of this analysis , 99 patients had discontinued the OLE study, with 28% of the patients discontinuing in the first year, and an annual discontinuation rate of 15 18% for existing patients in years 24 . Reasons for discontinuation are shown in . At study closure, of the 71 UC patients eligible to roll over into the phase 3 OLE study, 54 did so as a joint decision of patients and the treating investigators .
Patient disposition.
*In 2019, the sponsor made the decision to close the phase 2 TOUCHSTONE study after all active patients had completed at least 4 years of follow-up.
Of the 170 patients in the OLE, the mean age of patients at baseline for the double-blind study was 40.4 years, 57.6% of patients were male and 92.4% of patients were white . The mean duration of disease from diagnosis to the baseline of the double-blind study was 5.9 years, and most patients had not received prior anti-tumour necrosis factor therapy.
The mean exposure to ozanimod over the course of the study was 2.8 person-years .
Don’t Miss: Natural Enemas For Ulcerative Colitis